Kyle's BIMM 143 Portfolio

My classwork for BIMM143
Class 7: Machine Learning 1
Kyle Canturia (A17502778)
- Background
- K-means clustering
- Hierarchical Clustering
- Principal Component Analysis (PCA)
- Spotting major differences and trends
- PCA the rescue
- Digging deeper (variable loadings)
Background
Today we will begin our exploration of important machine learning methods with a focus on clustering and dimensionallity reduction.
To start testing these methods let’s make up some sample data to cluster where we know what the answer should be.
hist(rnorm(3000, mean = 10))
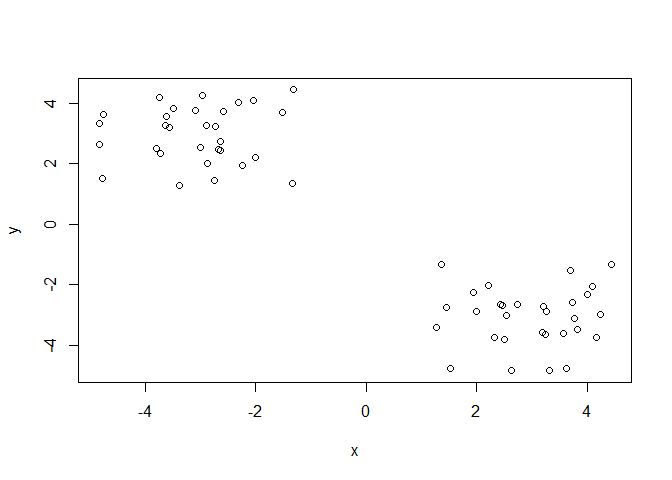
Q. Can you generate 30 numbers centered at +3 and 30 numbers at -3 taken at random from a normal distribution.
tmp <- c(rnorm(30, mean = 3),
rnorm(30, mean = -3))
x <- cbind(x=tmp, y=rev(tmp))
plot(x)
K-means clustering
The main function in “base R” for K-means clustering is called
kmeans(), let’s try it out:
k <- kmeans(x, centers = 2)
k
K-means clustering with 2 clusters of sizes 30, 30
Cluster means:
x y
1 -3.058139 2.961751
2 2.961751 -3.058139
Clustering vector:
[1] 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 1 1 1 1 1 1 1 1
[39] 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1
Within cluster sum of squares by cluster:
[1] 52.68442 52.68442
(between_SS / total_SS = 91.2 %)
Available components:
[1] "cluster" "centers" "totss" "withinss" "tot.withinss"
[6] "betweenss" "size" "iter" "ifault"
Q. What component of your kmeans result object has the cluster centers?
k$centers
x y
1 -3.058139 2.961751
2 2.961751 -3.058139
Q. What component of your kmeans result object has the cluster size (i.e. how many points are in each cluster)?
k$size
[1] 30 30
Q. What component of your kmeans result object has the cluster membership vector (i.e. the main clustering result: which points are in which cluster)?
k$cluster
[1] 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 1 1 1 1 1 1 1 1
[39] 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1
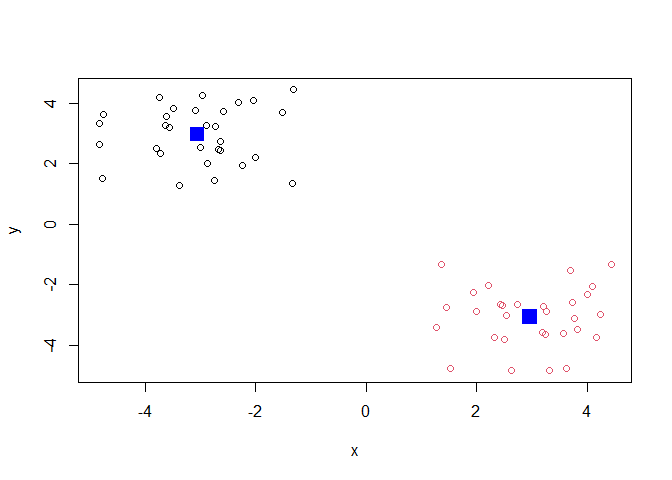
Q. plot the results of clustering (i.e. our data colored by the clustering result) along with the cluster centers.
plot(x, col=k$cluster)
points(k$centers, col="blue", pch=15, ,cex=2)
Q. Can you run
kmeans()again and clusterxinto 4 clusters and plot the results just like we did above with coloring by cluster and the cluster centers shown in blue?
j <- kmeans(x, centers = 4)
plot(x, col=j$cluster)
points(j$centers, col="blue", pch=15, cex=2)
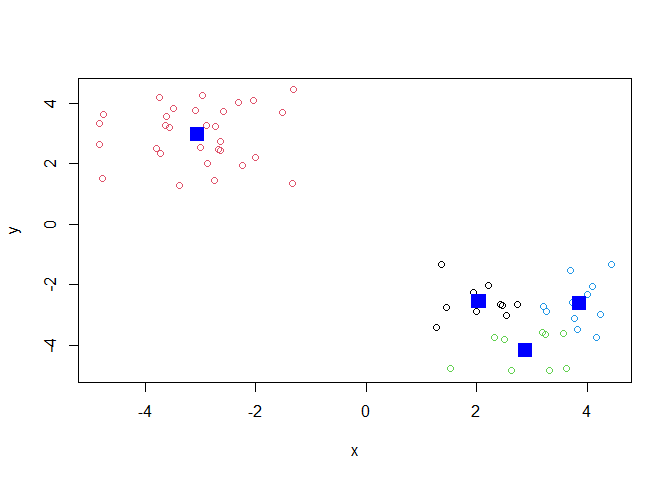
Key-point: Kmeans will always return the clustering that we ask for (this is the “K” or “centers” in K-means)!
k$tot.withinss
[1] 105.3688
Hierarchical Clustering
The main function for Hierarchical clustering in base R is called
hclust().
One of the main differences with respect to the kmeans() function is
that you cannot just pass your input data directly to hclust() - it
needs a “distance matrix” as input. We can get this from lot’s of places
including the dist() function.
d <- dist(x)
hc <- hclust(d)
plot(hc)
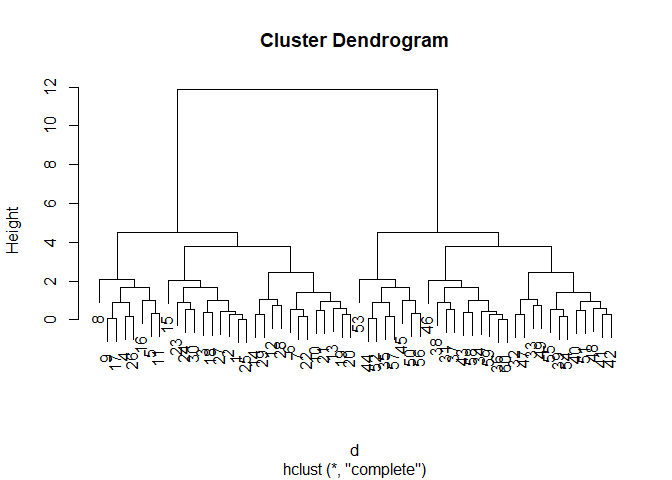
We can “cut” the dendrogram or “tree” at a given height to yield our
“clusters”. For this we use the function cutree()
plot(hc)
abline(h=10, col="red")
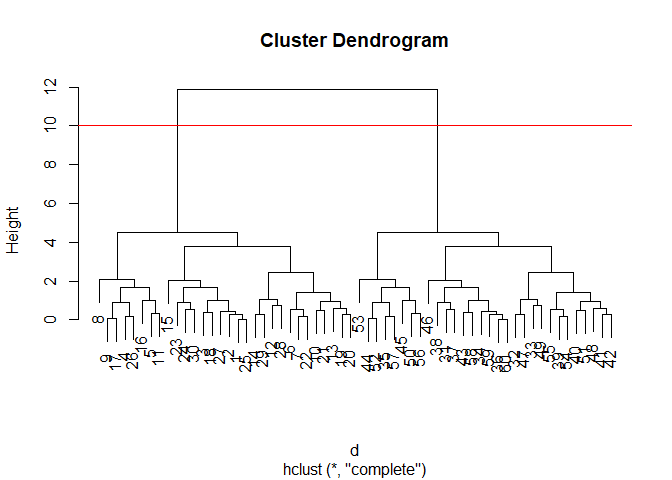
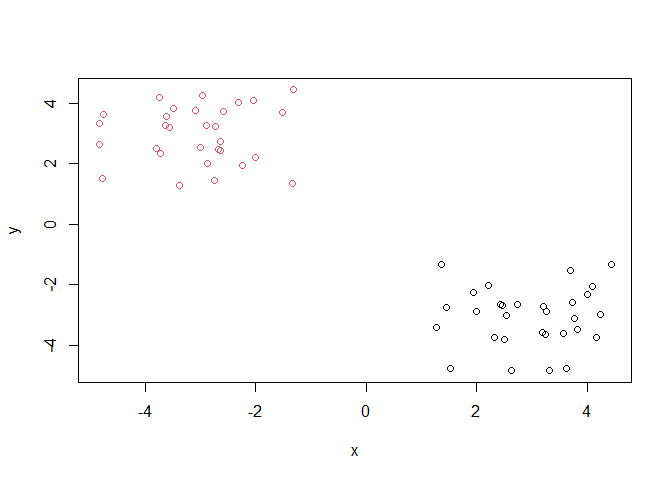
grps <- cutree(hc, h=10)
grps
[1] 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 2 2 2 2 2 2 2 2
[39] 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2
Q. PLot our data
xcolored by the clusterinv result fromhclust()andcutree()?
plot(x, col=grps)
Principal Component Analysis (PCA)
PCA is a popular dimensionality reduction technique that is widely used in bioinformatics.
##PCA of UK food data
Read data on food consumption in the UK
url <- "https://tinyurl.com/UK-foods"
x <- read.csv(url)
x
X England Wales Scotland N.Ireland
1 Cheese 105 103 103 66
2 Carcass_meat 245 227 242 267
3 Other_meat 685 803 750 586
4 Fish 147 160 122 93
5 Fats_and_oils 193 235 184 209
6 Sugars 156 175 147 139
7 Fresh_potatoes 720 874 566 1033
8 Fresh_Veg 253 265 171 143
9 Other_Veg 488 570 418 355
10 Processed_potatoes 198 203 220 187
11 Processed_Veg 360 365 337 334
12 Fresh_fruit 1102 1137 957 674
13 Cereals 1472 1582 1462 1494
14 Beverages 57 73 53 47
15 Soft_drinks 1374 1256 1572 1506
16 Alcoholic_drinks 375 475 458 135
17 Confectionery 54 64 62 41
It looks like the row names are not set properly. We can fix this.
rownames(x) <- x[,1]
x <- x[,-1]
x
England Wales Scotland N.Ireland
Cheese 105 103 103 66
Carcass_meat 245 227 242 267
Other_meat 685 803 750 586
Fish 147 160 122 93
Fats_and_oils 193 235 184 209
Sugars 156 175 147 139
Fresh_potatoes 720 874 566 1033
Fresh_Veg 253 265 171 143
Other_Veg 488 570 418 355
Processed_potatoes 198 203 220 187
Processed_Veg 360 365 337 334
Fresh_fruit 1102 1137 957 674
Cereals 1472 1582 1462 1494
Beverages 57 73 53 47
Soft_drinks 1374 1256 1572 1506
Alcoholic_drinks 375 475 458 135
Confectionery 54 64 62 41
A better way to do this is fix the row names assignment at import time:
x <- read.csv(url, row.names = 1)
x
England Wales Scotland N.Ireland
Cheese 105 103 103 66
Carcass_meat 245 227 242 267
Other_meat 685 803 750 586
Fish 147 160 122 93
Fats_and_oils 193 235 184 209
Sugars 156 175 147 139
Fresh_potatoes 720 874 566 1033
Fresh_Veg 253 265 171 143
Other_Veg 488 570 418 355
Processed_potatoes 198 203 220 187
Processed_Veg 360 365 337 334
Fresh_fruit 1102 1137 957 674
Cereals 1472 1582 1462 1494
Beverages 57 73 53 47
Soft_drinks 1374 1256 1572 1506
Alcoholic_drinks 375 475 458 135
Confectionery 54 64 62 41
Q1. How many rows and columns are in your new data frame named x? What R functions could you use to answer this questions?
dim(x)
[1] 17 4
nrow(x)
[1] 17
ncol(x)
[1] 4
17 rows, 4 columns.
Q2. Which approach to solving the ‘row-names problem’ mentioned above do you prefer and why? Is one approach more robust than another under certain circumstances?
I prefer the method we used that fixed the row names assignment at import time.
Spotting major differences and trends
Q3: Changing what optional argument in the above barplot() function results in the following plot?
Changing beside=T to beside=F.
Q4: Changing what optional argument in the above ggplot() code results in a stacked barplot figure?
Changing position = "dodge" to position = "stack".
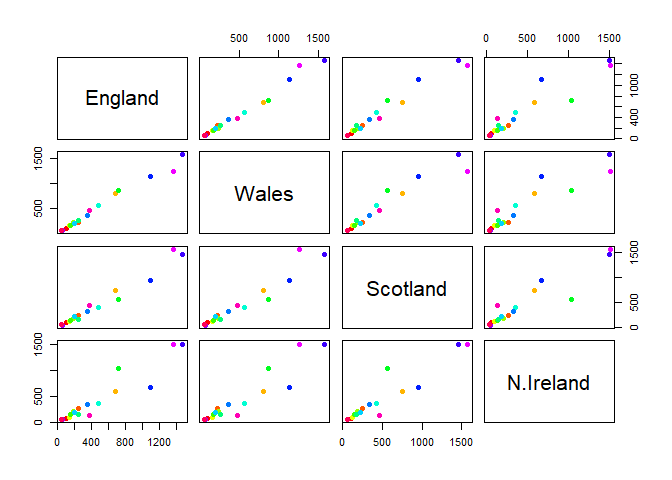
Q5: We can use the pairs() function to generate all pairwise plots for our countries. Can you make sense of the following code and resulting figure? What does it mean if a given point lies on the diagonal for a given plot?
pairs(x, col=rainbow(nrow(x)), pch=16)
library(pheatmap)
pheatmap( as.matrix(x) )
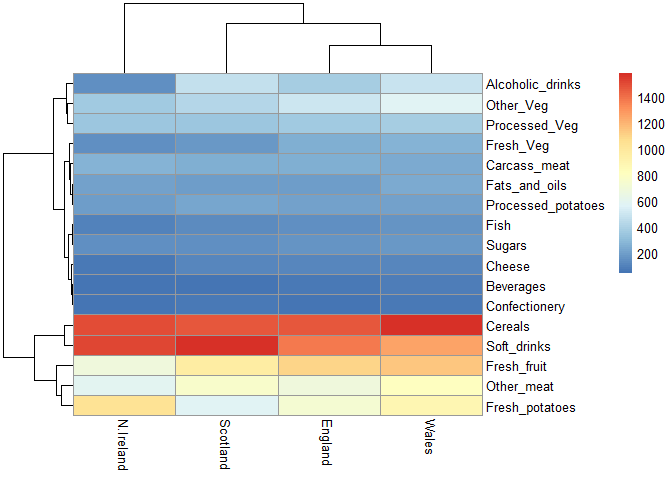
If a given point lies on the diagonal for a given plot, then the two countries match in terms of consumption for a given product. The heatmap shows higher consumption of a product with warmer colors, and lower consumption with cooler colors.
Q6: Based on the pairs and heatmap figures, which countries cluster together and what does this suggest about their food consumption patterns? Can you easily tell what the main differences between N. Ireland and the other countries of the UK in terms of this data-set?
For the most part, each country clusters together except for Northern Ireland reflecting similar food consumption between Scotland, England, and Wales. It’s a bit hard to tell, but they mainly differ on consumption of fresh fruit, other meat, and fresh potatoes. It’s definitely easier to see a mismatch on the pair plot rather than the heatmap figures, but there is still an observable difference on the heatmap.
Heatmap
We can install the pheatmap package with the install.packages()
command that we used previously. Remember that we always run this in the
console and not a code chunk in our quarto document.
Of all these plot really only the pairs() plot was useful. This
however took a bit of work to interpret and will not scale when I am
looking at much bigger datasets.
PCA the rescue
The main funciton in “base R” for PCA is called prcomp().
pca <- prcomp(t(x))
summary(pca)
Importance of components:
PC1 PC2 PC3 PC4
Standard deviation 324.1502 212.7478 73.87622 3.176e-14
Proportion of Variance 0.6744 0.2905 0.03503 0.000e+00
Cumulative Proportion 0.6744 0.9650 1.00000 1.000e+00
Q. How much variance is captured in the first PC?
67.4%
Q. How many PCs do I need to capture at least 90% of the total variance in the dataset?
Two PCs capture 96.5% of the total variance.
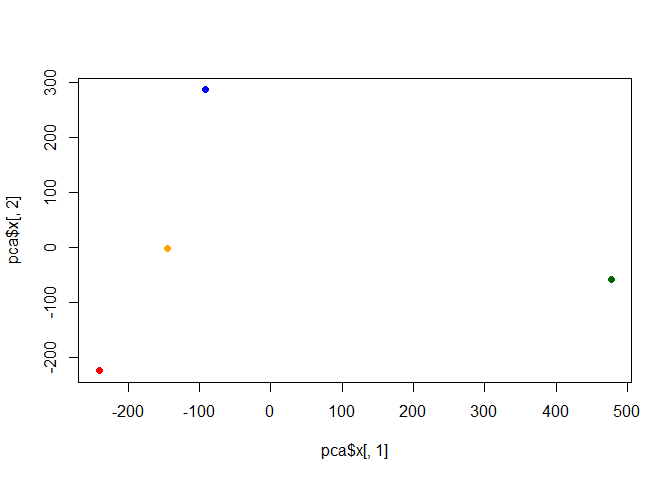
Q. Plot our main PCA result. Folks can call this different things depending on their field of study e.g. “PC plot”, “ordination plot”, “Score plot”, “PC1 vs PC2 plot”…
attributes(pca)
$names
[1] "sdev" "rotation" "center" "scale" "x"
$class
[1] "prcomp"
To generate our PCA score plot we want the pca$x component of the
result object
pca$x
PC1 PC2 PC3 PC4
England -144.99315 -2.532999 105.768945 -4.894696e-14
Wales -240.52915 -224.646925 -56.475555 5.700024e-13
Scotland -91.86934 286.081786 -44.415495 -7.460785e-13
N.Ireland 477.39164 -58.901862 -4.877895 2.321303e-13
my_cols <- c("orange", "red", "blue", "darkgreen")
plot(pca$x[,1], pca$x[,2], col=my_cols, pch=16)
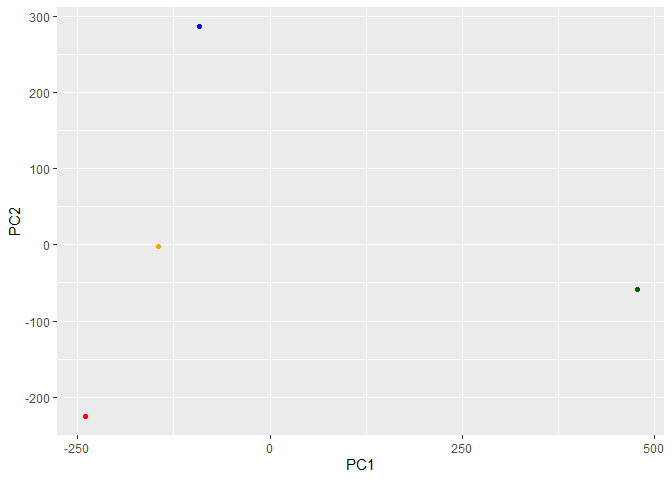
library(ggplot2)
ggplot(pca$x) +
aes(PC1, PC2) +
geom_point(col=my_cols)
Q7: Complete the code below to generate a plot of PC1 vs PC2. The second line adds text labels over the data points.
# Create a data frame for plotting
df <- as.data.frame(pca$x)
df$Country <- rownames(df)
# Plot PC1 vs PC2 with ggplot
ggplot(pca$x) +
aes(x = PC1, y = PC2, label = rownames(pca$x)) +
geom_point(size = 3) +
geom_text(vjust = -0.5) +
xlim(-270, 500) +
xlab("PC1") +
ylab("PC2") +
theme_bw()
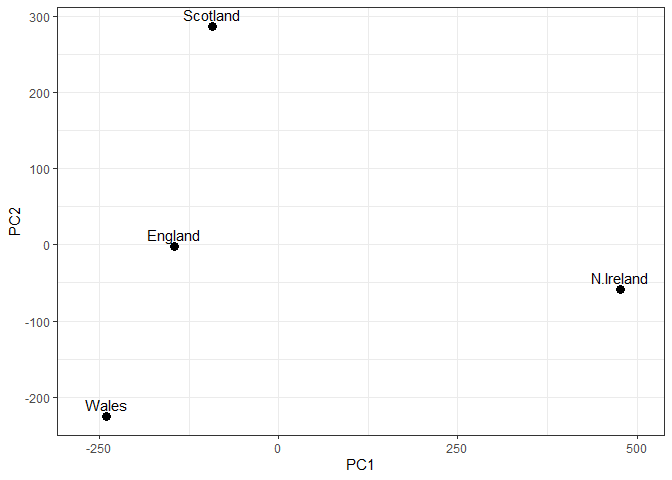
Q8: Customize your plot so that the colors of the country names match the colors in our UK and Ireland map and table at start of this document.
# Create a data frame for plotting
df <- as.data.frame(pca$x)
df$Country <- rownames(df)
# Plot PC1 vs PC2 with ggplot
colors <- c(Wales = "red", England = "tan", Scotland = "blue", N.Ireland = "darkgreen")
ggplot(pca$x) +
aes(x = PC1, y = PC2, label = rownames(pca$x)) +
geom_point(size = 3) +
geom_text(color = colors, vjust = -0.5) +
xlim(-270, 500) +
xlab("PC1") +
ylab("PC2") +
theme_bw()
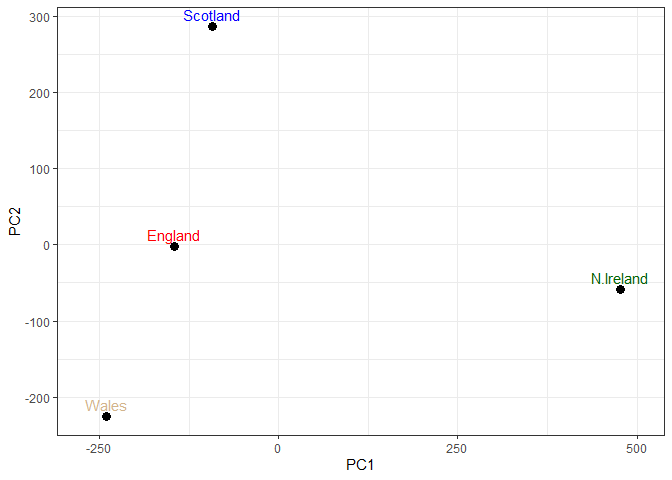
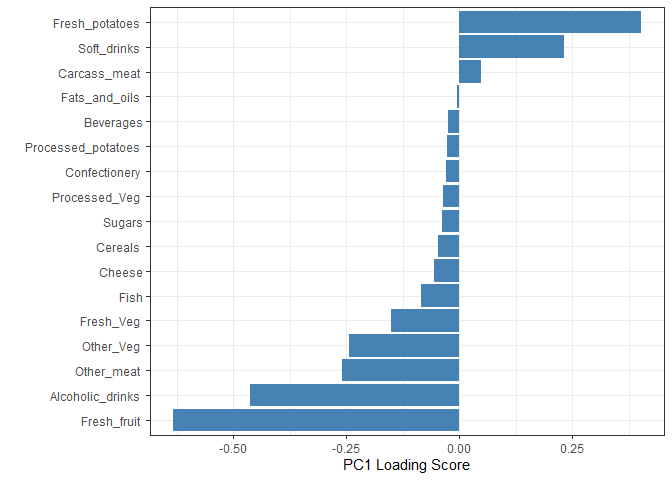
Digging deeper (variable loadings)
How do the original variables (i.e. the 17 different foods) contribute to our new PCs?
ggplot(pca$rotation) +
aes(x = PC1,
y = reorder(rownames(pca$rotation), PC1)) +
geom_col(fill = "steelblue") +
xlab("PC1 Loading Score") +
ylab("") +
theme_bw() +
theme(axis.text.y = element_text(size = 9))
Q9: Generate a similar ‘loadings plot’ for PC2. What two food groups feature prominantely and whoat does PC2 mainly tell us about?
ggplot(pca$rotation) +
aes(x = PC2,
y = reorder(rownames(pca$rotation), PC2)) +
geom_col(fill = "steelblue") +
xlab("PC2 Loading Score") +
ylab("") +
theme_bw() +
theme(axis.text.y = element_text(size = 9))
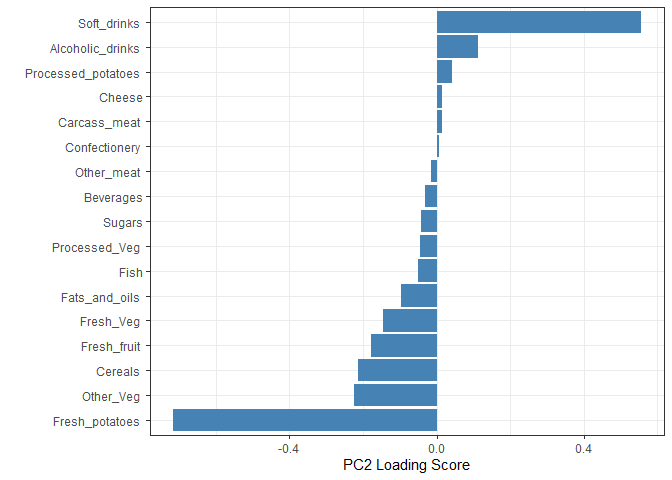
The two food groups that are the most prominent are soft drinks and alcoholic drinks. PC2 mainly provides information on the second highest variance in the data, after PC1.